Seqirus, a global leader in influenza prevention, will present new healthcare and cost outcomes data for its MF59®-adjuvanted trivalent influenza vaccine (FLUAD) at IDWeek in San Francisco (October 5-6). Results from five different studies conducted in two vulnerable populations – individuals 65 years and older and young children – show:
- Improved immune response in those 65 years and older associated with an increase in the dose of adjuvant MF591
- A significant reduction of 6% in all-cause hospitalization among elderly persons aged 65 years and above who live in U.S. nursing homes and received MF59-adjuvanted trivalent influenza vaccine compared to those who received a traditional egg-based trivalent vaccine2
- Cost-savings in using MF59-adjuvanted trivalent influenza vaccine vs. unadjuvanted trivalent vaccine for those 65 years+, where use led to a reduction in deaths and an incremental increase in quality-adjusted life years (QALY) 3
- Increased immunization against influenza among young children whose parents visited doctors receiving continued medical education (CME), with parents more than 30% more likely to have their child vaccinated against influenza, and children seen by the doctor with their parents 20% more likely to receive an adjuvanted trivalent influenza vaccine5
“The five studies presented at IDWeek reinforce the value of our MF59 technology in enhancing the body’s immune response, reducing the burden of influenza and providing greater protection for those whose weaker immune systems mean they are at an increased risk of influenza,” said Russell Basser, Senior Vice President of Research and Development at Seqirus.
MF59-Adjuvanted Influenza Vaccines in Individuals aged 65 years+ – Three Studies
A Phase 1 antigen and dose finding study, assessed the safety and immunogenicity of FLUAD in those 65 years of age and older. The results showed that increases in the dose of MF59 was associated with increased immunogenicity (immune response as measured by GMT) against all three strains contained in the vaccine (A/H1N1, A/H3N2, and B).1
In addition, researchers conducted a real-world evidence-based cluster randomized study where over 50,000 nursing home residents at more than 800 nursing homes were offered one of two influenza vaccines (MF59-adjuvanted trivalent vaccine or a traditional egg-based trivalent vaccine) to analyze the potential role of MF59-adjuvanted trivalent influenza vaccines in improving clinical outcomes. Pre-specified interim analysis with adjustments made for variables between the two sets of residents indicated a significant reduction (6%) in all-cause hospitalization with the MF59-adjuvanted vaccine compared to the population offered a traditional egg-based trivalent vaccine. Further, this reduced risk was observed during a season in which there was a reported reduced effectiveness of egg-based vaccines due to mismatch caused by egg-based mutagenesis.2
Another study, conducted in Argentina, evaluated the cost-effectiveness of MF59-adjuvanted trivalent influenza vaccine compared to an unadjuvanted vaccine. Based on this HECON model simulation, it is projected that replacement of unadjuvanted vaccine with MF59-adjuvanted trivalent influenza vaccine is likely to be a highly cost-effective intervention while also averting an estimated 530 deaths each season. 3
MF59-Adjuvanted Trivalent Influenza Vaccines in the Pediatric Population – Two Studies
A multicenter, randomized, controlled trial was conducted in Canada to determine whether an information, motivation and behavioral (IMB) skills model-based approach for continuing medical education (CME) or routine practice (no CME) would be effective in overcoming parental hesitancy of influenza vaccines. The CME addressed the burden of influenza in children, a range of barriers to vaccination along with an overview of available options. Immunization rates and HCP interactions with patients across the two physician groups were also reviewed. The results showed that parents seen by physicians in the CME group were more than 30 percent more likely to agree to immunize their child against influenza vs doctors in the routine practice group. Children who were seen along with their parents by doctors in the CME group were also more likely to receive an adjuvanted influenza vaccine vs those in the control group.5
A Phase III study conducted to evaluate the immunogenicity and safety of MF59-adjuvanted trivalent influenza vaccination among children 12 months through 6 years was utilized to assess the safety and immunogenicity of repeat vaccination. More than 600 subjects from the parent study6 were given a single dose of the MF59-adjuvanted trivalent influenza vaccine each influenza season for two consecutive seasons. The results showed similar safety outcomes to those of the Phase III study and improved immune response after repeat MF59-adjuvanted trivalent influenza vaccination compared to repeat non-adjuvanted influenza vaccination.4
“These data add to the body of evidence of the value of our adjuvanted seasonal vaccine among two at-risk populations,” said Gordon Naylor, Seqirus president. “MF59 technology continues to be a priority for us as part of our commitment to providing an influenza vaccine option that enhances protection against influenza as well as influenza-related outcomes.”
Poster Presentations at IDWeek
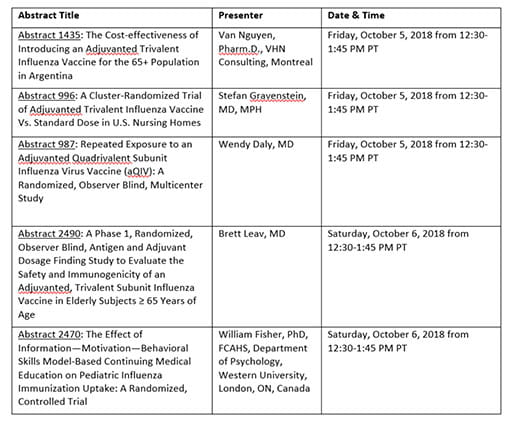
For more information, these five studies will be presented as poster presentations at the IDWeek conference:
About Seasonal Influenza
Influenza is a common, highly contagious infectious disease that can cause severe illness and life-threatening complications in many people. To reduce the risk of more serious outcomes, such as hospitalization and death, resulting from influenza, the CDC encourages annual vaccination for all individuals aged 6 months and older. Because transmission to others may occur one day before symptoms develop and up to 5 to 7 days after becoming sick, the disease can be easily transmitted to others. Influenza can lead to clinical symptoms varying from mild to moderate respiratory illness to severe complications, hospitalization and in some cases death. The CDC estimates that 600,000 people in the United States were hospitalized due to influenza-related complications during the 2016-2017 influenza season. Since it takes about 2 weeks after vaccination for antibodies to develop in the body that protect against influenza virus infection, it is best that people get vaccinated to help protect them before influenza begins spreading in their community.
About Seqirus
Seqirus is part of CSL Limited (ASX:CSL), headquartered in Melbourne, Australia. The CSL Group of companies employs more than 20,000 people with operations in more than 60 countries.
Seqirus was established on 31 July 2015 following CSL’s acquisition of the Novartis influenza vaccines business and its subsequent integration with bioCSL. As the second largest influenza vaccine provider in the world, Seqirus is a major contributor to the prevention of influenza globally and a transcontinental partner in pandemic preparedness.
Seqirus operates state-of-the-art production facilities in the US, the UK and Australia, and manufactures influenza vaccines using both egg-based and cell-based technologies, as well as a proprietary adjuvant. It has leading R&D capabilities, a broad portfolio of differentiated products and a commercial presence in more than 20 countries.
For more information visit www.seqirus.com and www.csl.com.
Media Contact
Polina.Miklush@Seqirus.com
+1 (908) 608-7170
Polina.Miklush@Seqirus.com
References
1. Leav B., Otten G., Ciralet M., et al. (2018) A phase 1, randomized, observer blind, antigen and adjuvant dosage finding study to evaluate the safety and immunogenicity of an adjuvanted, trivalent subunit influenza vaccine in elderly subjects ≥ 65 years of age. Presented at IDWeek 2018, October 2018.
2. Gravenstein S., Davidson H. E., Mcconeghy K., et al. (2018). A cluster-randomized trial of adjuvanted trivalent influenza vaccine vs. standard dose in U.S. nursing homes. Presented at IDWeek 2018, October 2018.
3. Nguyen V., Vizzotti C., Uruena A., et al. (2018). The cost-effectiveness of introducing an adjuvanted trivalent influenza vaccine for the 65+ population in Argentina. Presented at IDWeek 2018, October 2018.
4. Daly W., Ramsey K., Forsten A., et al. (2018). Repeated exposure to an adjuvanted quadrivalent subunit influenza virus vaccine (aQIV): a randomized, observer blind, multicenter study. Presented at IDWeek 2018, October 2018.
5. Fisher W., Gilca V., Murti M., et al. (2018). The effect of information—motivation—behavioral skills model-based continuing medical education on pediatric influenza immunization uptake: a randomized, controlled trial. Presented at IDWeek 2018, October 2018.
6. Vesikari T., Kirstein J., Devota Go G., et al. (2018). Efficacy, immunogenicity, and safety evaluation of an MF59-adjuvanted quadrivalent influenza virus vaccine compared with non-adjuvanted influenza vaccine in children: a multicenter, randomized controlled, observer-blinded, phase 3 trial. Lancet Resp Med, 6(5)345-356, doi:10.1016/S2213-2600(18)30108-5.
FLUAD® (Influenza Vaccines, Adjuvanted) Important Safety Information
Indication
FLUAD is an inactivated influenza vaccine indicated for active immunization against influenza disease caused by influenza virus subtypes A and type B contained in the vaccine. FLUAD is approved for use in persons 65 years of age and older.
Contraindications
Severe allergic reaction to any component of the vaccine, including egg protein, or after a previous dose of any influenza vaccine.
Warnings and precautions
- If Guillain-Barré syndrome (GBS) has occurred within six weeks of previous influenza vaccination, the decision to give FLUAD should be based on careful consideration of the potential benefits and risks.
- The tip caps of the prefilled syringes contain natural rubber latex, which may cause allergic reactions in latex-sensitive individuals.
Adverse reactions
- The most common (≥10%) local (injection site) adverse reactions observed in clinical studies were injection site pain (25%) and tenderness (21%).
- The most common (≥10%) systemic adverse reactions observed in clinical studies were myalgia (15%), headache (13%), and fatigue (13%).
For more information, please see accompanying US full Prescribing Information for FLUAD.
